The Varroa mite (Varroa destructor) originated in Southern Asia, where it has lived alongside the Asiatic Honeybee (Apis cerena) for millennia. Through the growth of international trade in the 20th century, Varroa was brought into contact with the European Honey bee (Apis mellifera) and has since caused insurmountable damage. Regardless of which of these species the Varroa mite is hosted by, its lifecycle remains much the same. Just as with Apis mellifera, varroa in Apis cerana colonies reproduce within capped brood cells, however the effect it has on these populations is minimal. This poses the question: what biological differences make Apis cerena so proficient at dealing with the Varroa mite?
Evolution has equipped Apis cerena with several defence traits against Varroa, which we will be exploring in the next few articles. One of the primary traits, and the one we delve into here, is ‘hygienic behaviour’ and altruistic suicide.
Honey bees are an example of a Eusocial insect, with highly organized social structures and defined roles such as reproduction, brood care and foraging. Although both Apis cerena and Apis mellifera are able to recognize and remove infected, wounded or dead brood from capped cells, this practice, known as ‘hygienic behaviour’, is far less common with Apis mellifera. Apis cerena on the other hand remove infected or damaged brood almost immediately, very often before the brood even perishes. To trigger hygienic behaviour, damaged brood emit a chemical signal that worker bees sense and respond to. This defence mechanism stops, which results in the death of the brood, also stops the potential spread of varroa, attaining social immunity for the colony.
In the 2016 study ‘Social apoptosis in honey bee superorganisms’, researchers compared the impact of varroa on both Apis cerana and Apis mellifera brood, and the effect of hygienic behaviour on the colonies. To observe Varroa’s effect on brood development, they placed infested and non-infested control brood from both species in an incubator. This controlled environment was to imitate the hive, without the interference of worker bees. Cells were then uncapped one day prior to the expected emergence date, and the specimens analysed. In Apis mellifera, both infested and non-infested brood were similarly developed. However, in Apis cerana, infested brood were heavily damaged and under developed compared to the control groups.
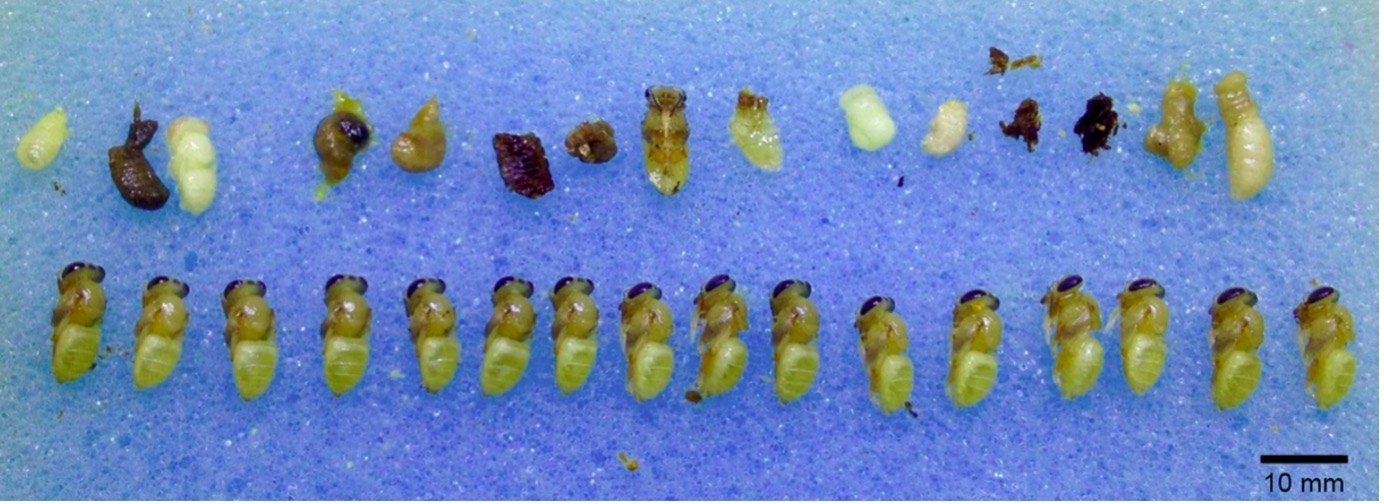
Fig. 1: Mite-infested brood (above row) and control brood (bottom row) from an A. cerana colony (Phatthalung, Thailand). Individuals were removed from their cells one day before emergence was expected. Most infested individuals stopped development at the larval and prepupal stages and died, Photo by Paul Page.
To take this further, researchers delivered small benign wounds to samples of Apis cerena and Apis mellifera larvae and placed them in an incubator and their respective colonies. In both environments, Apis cerana larvae died faster and in greater proportions compared to Apis mellifera, despite receiving the same wounds. Further more, Apis cerana workers removed wounded brood far more often and much faster than Apis mellifera, demonstrating a higher sensitivity of worker bees to dysfunctional brood.
Researchers concluded that the effective responsiveness of Apis cerena is directly caused by the brood of this species being significantly weaker, coupled with a higher sensitivity of worker bees toward damaged brood. Paradoxically, being more vulnerable to varroa may in fact be a part of the colonies defence mechanism against it. As Apis cerana brood are more fragile, the chemical signal is triggered far sooner and stronger than that of Apis mellifera. Workers, who are also more sensitive, can react faster and have a better chance of interfering with the spread of the parasite. In this way, Apis cerana is behaving like a multi-cellular organism. This is the process of ‘Apoptosis’, where sick or damaged cells trigger a self-destruct mechanism, in a sense committing ‘altruistic suicide’, and in doing so, stopping the spread of infection to protect the organism as a whole.
References
Social apoptosis in honey bee superorganisms
Paul Page, Zheguang Lin, Ninat Buawangpong, Huoqing Zheng, Fuliang Hu, Peter Neumann, Panuwan Chantawannakul & Vincent Dietemann, 2016
https://www.nature.com/articles/srep27210#:~:text=Altruistic%20suicide%20of%20immature%20bees,social%20immunity%20in%20honey%20bees.